Technology
Reducing Costs & Improving Outcomes
pCT from Proton Calibration Technologies is an integrated workflow solution that ameliorates a significant issue for proton therapy centers. The clinical need is to efficiently plan and deliver a high-quality treatment safely using large doses and fewer sessions. Join us on our mission to commercialize the world’s fifth medical imaging modality leveraging our precise, proven, proprietary, and profitable pCT technology.
Technology
Proton Computed Tomography (pCT)

Contact
Proton Calibration Technologies
986 N. Cedar Cove Road
Hartselle, AL 35640
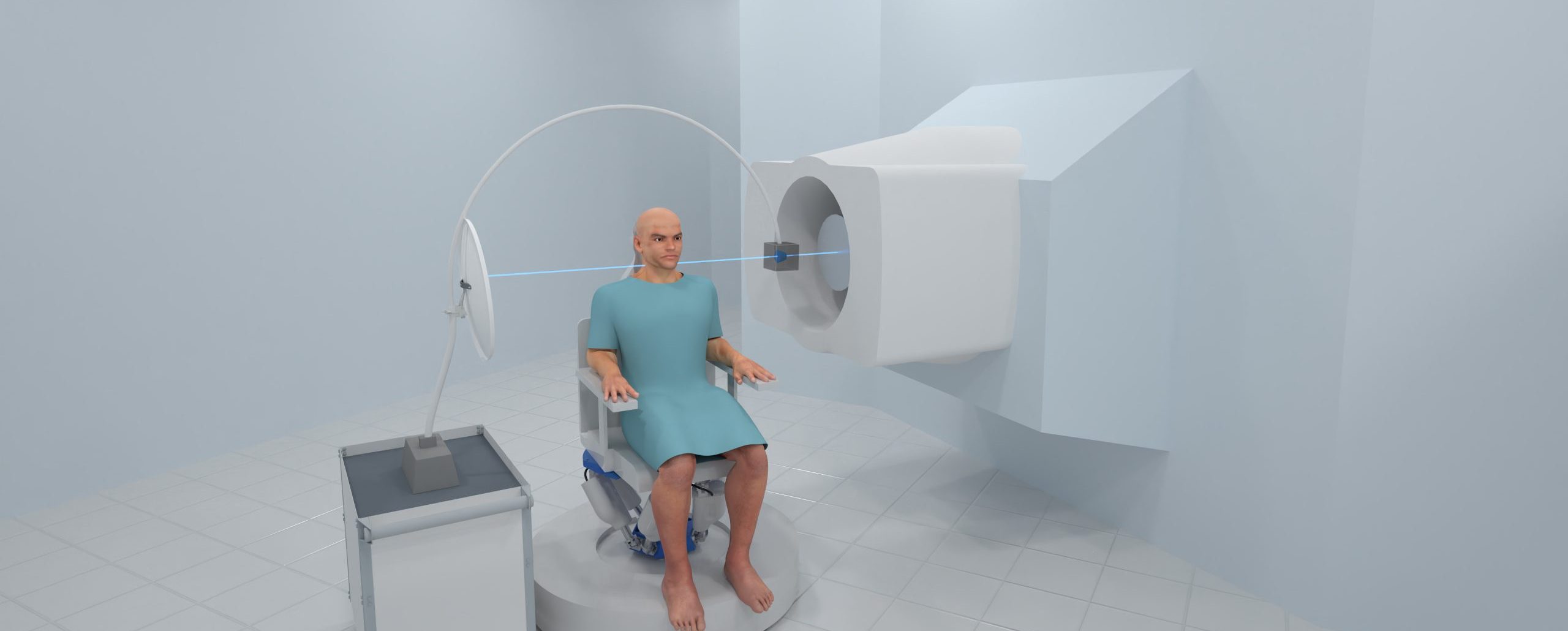
How a 3D pCT Scan is Done
The video below shows how a proton Computed Tomography (pCT) scan is done.
The purpose of any scan is to collect data. The data is then sent to an algorithm, that is, a software program to create the three dimensional image of a region of interest in the patient.
The data is obtained by sending beams through the patient and recording how the beams are changed by the tissue they encounter. Beams consist of packets of protons traveling at very high speed. Every proton is measured separately, resulting in crisp images and low total dose to the patient.
In the future there will be non-medical uses for pCT. We have already had discussions with satellite shielding and tank armor research centers.
How Tumors are Treated with Protons
The video below shows how patients are treated with protons. The video was created by ProTom International, which has graciously provided permission for us to present it.
Our proton Computed Tomography (pCT) imaging system, will work with any proton therapy vendor’s system, but would work particularly well with ProTom’s system. ProTom’s system provides the highest beam energy of any commercial proton therapy system. The higher the beam energy used for a pCT scan, the sharper the image and the lower dose to the patient.
We look forward to working with vendors, such as ProTom, to integrate our pCT system into the overall patient treatment planning process.